Last Publications:
Ouedraogo DW, Lenck-Santini PP, Marti G, Robbe D, Crépel V, Epsztein J. Abnormal UP/DOWN Membrane Potential Dynamics Coupled with the Neocortical Slow Oscillation in Dentate Granule Cells during the Latent Phase of Temporal Lobe Epilepsy. eNeuro. 2016 May 31;3(3).
Artinian J, Peret A, Mircheva Y, Marti G, Crépel V. Impaired neuronal operation through aberrant intrinsic plasticity in epilepsy. Ann Neurol. 2015 Jan 13.
Crépel V, Mulle C. Physiopathology of kainate receptors in epilepsy. Curr Opin Pharmacol. 2015 Feb;20:83-8.
Peret A, Christie LA, Ouedraogo DW, Gorlewicz A, Epsztein J, Mulle C, Crépel V. Contribution of aberrant GluK2-containing kainate receptors to chronic seizures in temporal lobe epilepsy. Cell Rep. 2014 Jul 24;8(2):347-54.
INSERM Transfert filed patents:
– Method and pharmaceutical composition for use in the treatment of epilepsy (V. Crépel, C. Mulle, A. Peret; WO/2015/036618)
– Methods and pharmaceutical composition for the treatment of post-traumatic epilepsies (C. Rivera, V. Crépel, C. Pellegrino, N. Kourdougli; EP15306384)
Scientific Interests
We investigate neuronal computation and plasticity in normal and pathological conditions. Our studies are conducted at multiscale levels i.e. from the individual spine to the microcircuit.
Disease conditions: Temporal Lobe Epilepsy and Traumatic Brain Injury.
Key words: hippocampus, cerebellum, synapse, intrinsic property, plasticity, synaptopathy, calcium
Technical skills: (i) in vitro and in vivo electrophysiological recordings; (ii) calcium imaging; (iii) morphometric analysis; (iv) optogenetic; (v) spatial navigation in virtual reality environments.
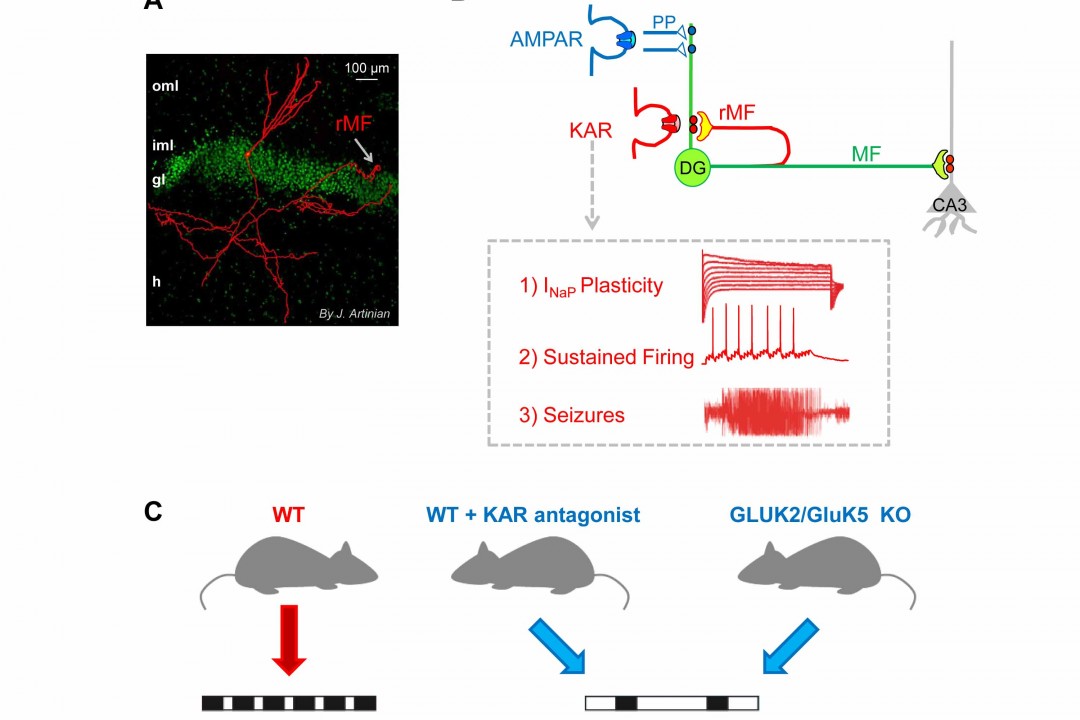
Kainate receptors in the physiopathology of temporal lobe epilepsy
Temporal Lobe Epilepsy (TLE) is the most common form of partial epilepsy in adults is often refractory to pharmacological medication. Moreover, patients with TLE also often suffer from comorbid disorders including cognitive impairments. Therefore, it is crucial to better understand the physiopathology of TLE in order to propose new anti-epileptic strategies and shed light on putative alterations of neuronal computation.
We show that recurrent mossy fibers synapses operate via kainate receptors not present in naïve conditions (Epsztein et al. 2005). These synapses profoundly affect synaptic transmission by generating synaptic events with slow kinetics. Because of this feature, we show that KAR-operated synapses impose an aberrant synaptic temporal integration, a wrong tempo of firing and trigger a long-lasting intrinsic plasticity in epileptic dentate granule cells (Epsztein, Sola et al. 2010; Artinian et al. 2011; Artinian et al 2015). We propose that these changes of neuronal computation may contribute to the impairment of dentate gyrus functions such as gate function and pattern separation in TLE.
In collaboration with C. Mulle (IINS, Bordeaux), we recently demonstrate that GluK2/GluK5 kainate receptors ectopically expressed in dentate granule cells play a major role in recurrent seizures in TLE (Peret, Christie et al. 2014;Crepel and Mulle 2014). This should revitalize the development of new pharmacological agents and strategies directly targeting these KARs as novel antiepileptic drugs: INSERM Transfert filed patent: WO/2015/036618.
Collaborators
INMED
Dr. C Rivera
Dr. R. Khazipov
Dr. R. Cossart
Dr. A. Torcini
Dr. J. Epsztein
Dr. PP Lenck-Santini
Dr. I. Bureau
Dr. Represa
EXTERNAL
Dr. C. Mulle (Institut des Neurosciences, Bordeaux)
Pr. F. Bartolomei (Hopital Timone, Marseille)
Pr. D. Scavarda (Hopital Timone, Marseille)
Dr. Jean-François Perrier (Copenhagen, Danemark)
Dr. M. Simonneau (Centre de Psychiatrie & Neurosciences, Paris)
Former members
– Jérôme Epsztein (CR1, INSERM)
– Thomas Scalfati (Master Student)
– Saara Valpurla (Master Student, Erasmus)
– Julien Artinian (Post-doc.)
– Geoffrey Marti (Master Student)
– Louisa Christie (Post-Doc.)
– David Ouedraogo (PhD student)
– Yanina Mircheva (Master Student)
– Karen Arnaud (Master Student)
Funding
2013 – 2017 : ANR « Blanc » (TRAUMEP, C. RIVERA & V. CREPEL)
2013-2014: LFCE (A. PERET, V. CREPEL)
2011 : région PACA : Appel à Projets Ouverts (projet APO, V. CREPEL)
2011-2012: LFCE (J. ARTINIAN, V. CREPEL)
2010 – 2014: ANR « Blanc » (KAREP, C. MULLE & V. CREPEL)
2010 FRC / Rotary (A. REPRESA & V. CREPEL)
2009 – 2012: ANR « Blanc » (Epileptic Code, V. CREPEL)
2006 – 2009: ANR MNP (CDDRat, A. REPRESA & V. CREPEL)
Join our team !
Our team has open positions for master students, PhD students and post-docs. Please send your application by email to Valérie Crépel